Who knew that in March, a novel virus had the means to impact our lifestyle. In less than a year we would also have a vaccine to solve this pandemic. We have learned about how clinical trials works, and along with the promising results from the initial set of vaccines. Now comes the hard part – getting these vaccines distributed. This sounds simple, but these novel vaccines come with logistical issues. Leading up to extra burden on hospitals.
In this blog post I will look to cover the following:
- Does Operation Warp Speed cover the complete Vaccine Supply Chain?
- How will Hospitals manage the storage, and monitoring of these Vaccines (as per the CDC guidelines)
- Can the Internet of Things (IoT) devices solve the logistics issues for these vaccines?
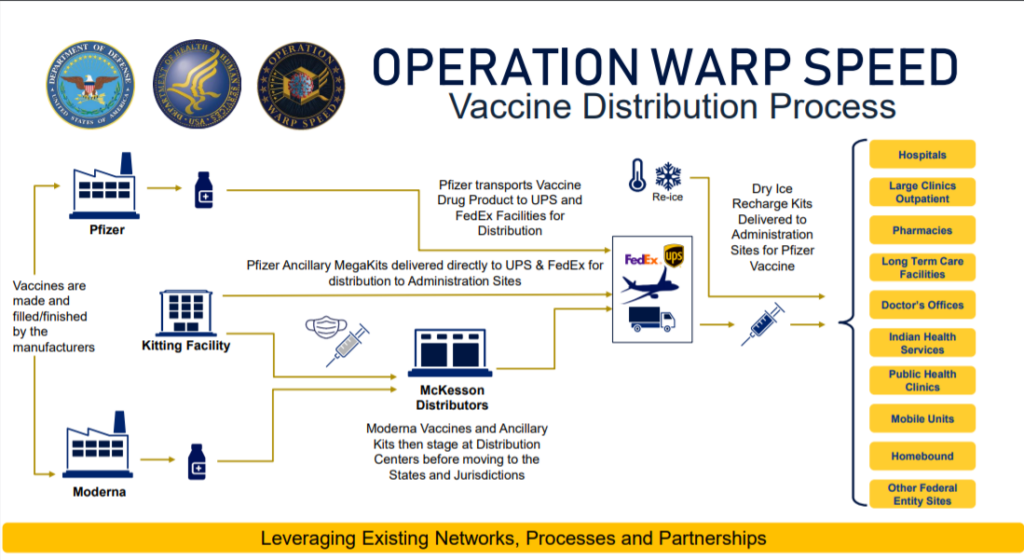
The Government effort behind getting these new vaccines to the public is tremendous. The typical vaccine development (from design through clinical trials) has changed. In this diagram you see the two initial vaccine candidates from Pfizer and Moderna. Both companies have applied for ‘early use authorization’ (EUA) from the FDA.
The key takeaway is that the shipment monitoring stops at the distribution sites. So the responsibility for managing these vaccines on the hospitals.
Not all vaccines are the same
Another key question is about refrigeration for these vaccines. There are three kinds of refrigeration:
- Refrigeration from 2 to 8 degree C
- Frozen from -15 to -25 degree C
- Ultra cold from -60 to -80 degree C
Let’s take the Pfizer vaccine temperature need. Shipments will be in the ultra cold range. So there is a limited number of locations that can support temperature requirements, and this impacts who can received these vaccines (rural versus urban hospitals).
If we look at Moderna’s temperature need. We can use devices to do ‘temperature’ monitoring. Why do this? Because we can ensure the the equipment used doesn’t fail, and the product doesn’t degrade.
What the CDC recommends
The CDC has issued guidelines for how these vaccines are to be managed.
Here are the key points:
- “for a reliable ‘cold chain.’ 3 elements must be in place
- a well-trained staff
- reliable storage and temperature monitoring equipment
- accurate vaccine inventory management” page 33
- “every vaccine storage unit/container must have a temperature monitoring device. CDC recommends a digital data logger (DLLs).” page 33
- “if COVID-19 vaccines are exposed to temperature excursions at any time, the temperature excursion should be documents and reports according to the jurisdiction immunization program’s procedures. The vaccines that were exposed to ‘out-of-range’ temperatures must be labeled “do not use” and stored at the required temperature until further information on usability can be gathered…” page 34
You can find the CDC COVID-19 Vaccination Program Interim Playbook … as of October 29, 2020
There is a lot for hospitals to manage from the CDC guidelines.
IoT helps solve the COVID-19 vaccines storage dilemma
I have been an advocate for the use of IoT for the US Pharmaceutical supply chain. I have provided feedback to the FDA on the Drug Supply Chain Security Act (DSCSA). The past few years I’ve applied Roambee IoT devices for ‘track and trace.’ As well as clinical trial supply management (in both the US and EU regions). So the challenge for these vaccines is the need for ‘track and trace,’ and temperature monitoring.
There are materials challenges in monitoring the temperature of ultra-cold refrigeration. IoT devices can meet the test of ‘frozen’ temperatures. Which is ideal for the Moderna, and later vaccines, down to -35C. We have tested IoT devices in freezers and will present these results at an IoT event later this month (refer to the link towards the end of this post).
Benefits for the Hospitals
We know that the hospitals have their hands full with treating COVID patients. Larger hospitals may have available staff to do the daily monitoring and reporting. So what are the benefits for hospitals using IoT devices?
- The temperature data shows we can measure vaccines in freezers. They can report out temperature information. Any deviation will send out a text message and email alert to the hospital staff.
- These devices will alert when the refrigerator door is open (via a built-in light sensor). We hope the hospitals have a level of security, so that only key personal have access to these vaccines. If not these devices can identify if an unauthorized person opens the door.
- Because the IoT sensors are ‘inside’ the refrigerator, and operate on battery power. In the event of a power outage they will continue to operate and record temperature data. This data may prove critical if the power is out for prolonged period of time.
- Another benefit is IoT communications. Typical hospital locations for these freezers are in enclosed rooms with no windows. The results of our testing shows that these devices work well.
- Using IoT devices would improve vaccine quality. They provide a systems for maintaining these vaccines at their temperature specs.
Call to Action
Was this post helpful for you? We will be presenting at the Grand Slam IoT Community event on December 11th. We will go into further detail (test results) on how this will work for Hospitals and the COVID-19 vaccines.
Session title: COMPLETE COVID-19 COLD CHAIN SOLUTION FOR A HOSPITAL
Virtually yours,
Jim
Sources
Cecere, Lora. “The Economy Needs A Shot In The Arm. The Question Is How.”. Forbes, 2020, https://www.forbes.com/sites/loracecere/2020/11/19/the-economy-needs-a-shot-in-the-arm-the-question-is-how/?sh=f950d995e3ba. Accessed 5 Dec 2020.
“Rural Hospitals Can’t Afford Freezers To Store A Covid-19 Vaccine – STAT”. STAT, 2020, https://www.statnews.com/2020/11/11/rural-hospitals-cant-afford-freezers-to-store-pfizer-covid19-vaccine/. Accessed 5 Dec 2020.